In summary, effective document control is essential for ensuring the accuracy and consistency of important information and for preventing errors and misunderstandings. By implementing the components outlined above, organizations will be on the way to maintaining the integrity of their information. For more information on how Accupoint Software can help streamline your document control process, we invite you to schedule a platform demonstration today.
Proper calibration of inspection tools is essential for ensuring the accuracy and reliability of measurement results. By following these best practices, you can ensure that your inspection tools are operating within their specified tolerance limits and that the measurements taken are accurate and repeatable. For more information on how Accupoint Software can help streamline your calibration process, please contact us today to discuss your requirements.
1. Plan, Do, Check, Act
The Plan, Do, Check, Act (PDCA) cycle is a widely-used method for achieving continual improvement of quality management systems. The PDCA cycle consists of four steps; Plan, where organizations establish clear goals and objectives for their QMS; Do, where the organization implements the plan; Check, where the organization reviews and evaluates the QMS; and Act, where the organization implements corrective and preventative actions to improve the QMS. By continuously repeating the PDCA cycle, organizations can identify and address issues with their QMS, and continuously improve it in order to meet customer needs and achieve organizational goals. 2. Six Sigma Six Sigma is a methodology that can be used to achieve continual improvement of quality management systems. Six Sigma is a data-driven approach that focuses on identifying and eliminating defects in processes in order to achieve near-perfect quality. Six Sigma uses a variety of tools and techniques, such as statistical process control and process mapping to identify and eliminate sources of variation in processes. By implementing Six Sigma, organizations can improve the efficiency and effectiveness of their processes and ultimately improve customer satisfaction and achieve organizational goals. 3. Lean Lean is a practice that focuses on eliminating waste and improving efficiency in processes. Lean uses a variety of tools and techniques, such as value stream mapping to identify and eliminate sources of waste in processes. By implementing Lean, organizations can reduce costs, improve quality and increase customer satisfaction. Lean also encourages teams to continuously identify and eliminate waste in their processes. Lean Six Sigma is a combination of both Lean and Six Sigma which can provide more comprehensive improvement in a business process. 4. Total Quality Management TQM is a management approach that involves all employees in continuously improving the processes and systems that produce goods and services. TQM focuses on customer satisfaction, employee involvement, and continuous improvement. One of the key principles of TQM is that quality is everyone’s responsibility, which means that all employees should be involved in the quality management system and continuously work towards improving it. TQM also emphasizes the use of data and facts to guide decision making and problem solving. TQM provides a comprehensive approach that covers all aspects of the business and requires regular review, evaluations, and improvement to ensure it is keeping up with the changing needs of the business. To learn more about how Accupoint Software can help with your continual improvement activities, please contact us today to arrange for a discovery session. A strong corrective and preventive action procedure is one of the most important, and value-added processes, in an effective quality management system. Despite the benefits, many oil and gas companies fail to have robust procedures in place or have significant gaps that contribute to audit findings. In our experience, the most common mistakes fall into the following categories:
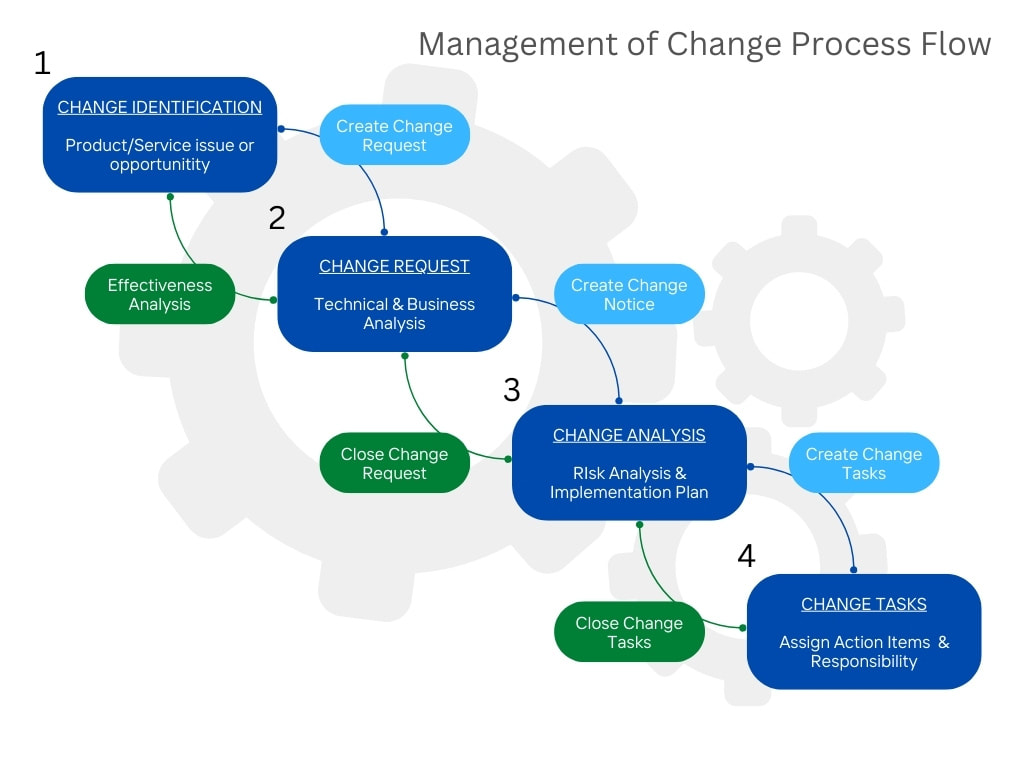
1. No Documented CAPA Procedure The CAPA procedure is one of the most important procedures in your quality management system. Despite this fact, many oil and gas companies have no formalized procedure for the management of corrective and preventative actions. 2. Failure to verify the effectiveness of the CAPA Another common mistake is to close the CAPA before verifying or validating the effectiveness of the action(s). 3. Procedures for CAPA are not adequately established Some oil and gas companies have a CAPA procedure in place but are missing one or more of the key process steps. 4. The CAPA process is totally managed by the Quality Department Use a cross functional team to work on your CAPAs. 5. Every non-conformance is turned into a CAPA Not every nonconformance requires a CAPA. Overburdening your CAPA system will result in loss of effectiveness and team focus. 6. No root cause analysis Root cause analysis is a vital step in the corrective action process and is overlooked or not performed at all. 7. Inadequate effectiveness checks Make sure to have a specific and well-planned system to verify the effectiveness of corrective actions. For more information on how Accupoint can help you to streamline your corrective and preventive action program, please contact us today. We’re here to help. In talking with clients and prospects, one of the major challenges that we hear over and over relates to management of change. Many organization struggle with the requirement, resulting in a broken system and ultimately a nonconformance. In drilling down on the issues, we often discover the root cause is related to an inefficient process. Specifically, failures in focus, expediency, communication, and coordination across the organization. At Accupoint Software, we have designed a comprehensive solution to deliver consistent and predictable performance across the enterprise. The following flow chart illustrates our 4 step approach to ensure control of your management of change process. If you are interested in learning how Accupoint can help with your management of change program, we invite you to contact our team today.
|
Archives
May 2024
|