|
A strong corrective and preventive action procedure is one of the most important, and value-added processes, in an effective quality management system. Despite the benefits, many oil and gas companies fail to have robust procedures in place or have significant gaps that contribute to audit findings. In our experience, the most common mistakes fall into the following categories:
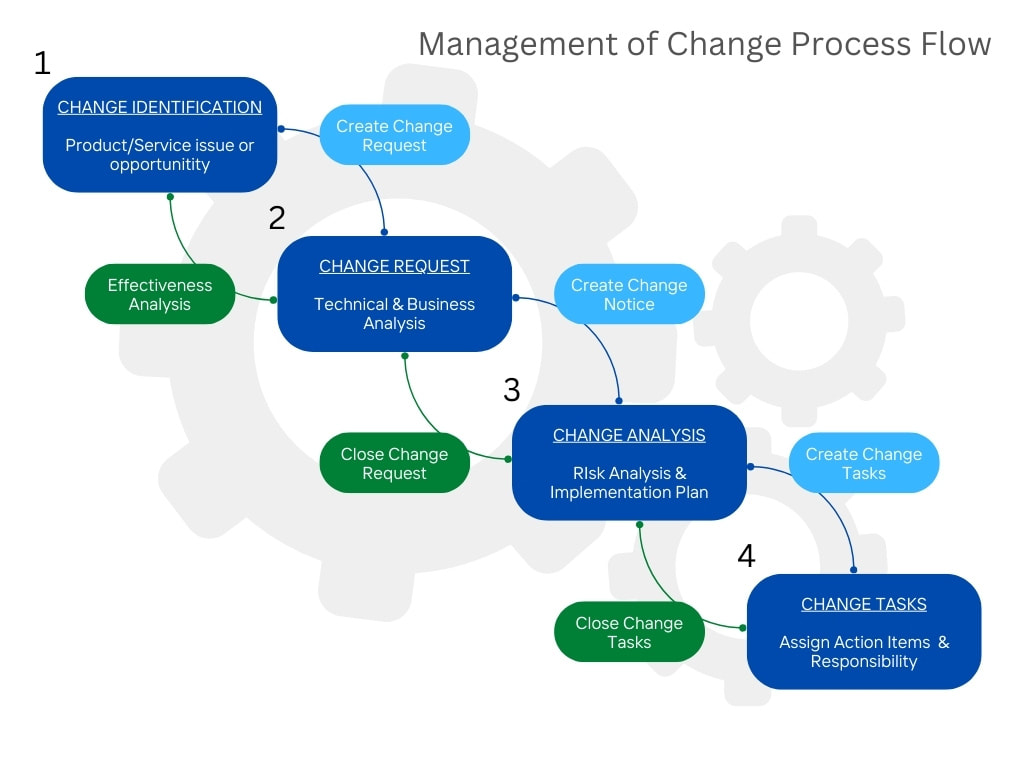
1. No Documented CAPA Procedure The CAPA procedure is one of the most important procedures in your quality management system. Despite this fact, many oil and gas companies have no formalized procedure for the management of corrective and preventative actions. 2. Failure to verify the effectiveness of the CAPA Another common mistake is to close the CAPA before verifying or validating the effectiveness of the action(s). 3. Procedures for CAPA are not adequately established Some oil and gas companies have a CAPA procedure in place but are missing one or more of the key process steps. 4. The CAPA process is totally managed by the Quality Department Use a cross functional team to work on your CAPAs. 5. Every non-conformance is turned into a CAPA Not every nonconformance requires a CAPA. Overburdening your CAPA system will result in loss of effectiveness and team focus. 6. No root cause analysis Root cause analysis is a vital step in the corrective action process and is overlooked or not performed at all. 7. Inadequate effectiveness checks Make sure to have a specific and well-planned system to verify the effectiveness of corrective actions. For more information on how Accupoint can help you to streamline your corrective and preventive action program, please contact us today. We’re here to help. In talking with clients and prospects, one of the major challenges that we hear over and over relates to management of change. Many organization struggle with the requirement, resulting in a broken system and ultimately a nonconformance. In drilling down on the issues, we often discover the root cause is related to an inefficient process. Specifically, failures in focus, expediency, communication, and coordination across the organization. At Accupoint Software, we have designed a comprehensive solution to deliver consistent and predictable performance across the enterprise. The following flow chart illustrates our 4 step approach to ensure control of your management of change process. If you are interested in learning how Accupoint can help with your management of change program, we invite you to contact our team today.
Send us your data and our team will configure your workspace. With our Legacy Load™ service, we take the hassle out of converting to a new system. Our support team will oversee loading all of your data, users, and related information into a workspace, and alert you when it’s ready for use. In addition, we will provide your team with in-depth training to outline platform features and functionality.
At Accupoint Software, we provide oil & energy companies with hassle free solutions to manage their quality and compliance management programs. Contact us today to find out more about how we can simplify your move to a digital quality management system.
As such, here are five components that are integral to a successful quality management system:
The ability to access and then make decisions based on up-to-date information is one of the best ways to demonstrate to customers the steps you are taking to improve your product quality and delivery performance. You will find this functionality and more contained in Accupoint Software’s intuitive web-based quality management platforms. With Accupoint, you can achieve sustainable improvements to cost, efficiency, up-time, safety, and profitability. For more information on how Accupoint Software can help positively impact customer perception, we invite you to contact us today to discuss specifics.
What is involved in the document approval process?
It depends. Oil and energy companies go about formalizing and approving documents in many ways. It could be as easy as defining the steps and processes that need to be completed. Typically, that is the initial step in most programs, and with automation software, that aspect can be more efficient and streamlined, enabling document approval to be implemented and communicated in a timely fashion. A document normally goes through a series of approval steps, all of which can benefit from automation. The first step of this process is the document’s submission for approval. Next, people will be assigned to review the document for completeness and determine if it meets the standard for approval. Each person assigned to the approval process must assigned an applicable permission level, and timeframes should be utilized to guarantee that the process moves forward in an expedited fashion. How to design, create and streamline the document approval process When designing your document approval process, there are a number of paths you could take based on internal variables. Thus, you will want to consider the following items to create and optimize your document approval process:
Final thoughts Document approval is an integral part of an effective document management program. While it is an important process, it can be tedious and time-consuming. With an integrated QMS software like Accupoint, you will get a simple-to-use, intuitive system that combines the latest cloud technology and industry best practice to guarantee that your document control process meets or exceeds the requirements of international compliance standards such as API Spec Q1, API Spec Q2 or ISO 9001. Contact us today to learn today how Accupoint can help streamline your document approval process. |
Archives
May 2024
|